Is consensus secretly killing your AI pipeline?
Why you need to abandon old data models and slow decisions today.
HT4LL-20260428
Hey there,
Waiting for the perfect animal model, the perfect data privacy approval, or the perfect committee consensus is the absolute fastest way to kill your pipeline in the AI era.
As R&D leaders, we are constantly battling the friction between innovation and legacy infrastructure. We are stuck relying on single-group digital health studies, 90% clinical failure rates driven by fundamentally flawed animal models, and data privacy bottlenecks that stall critical research for months. Worse, when we try to implement cutting-edge AI or wearables to solve these very issues, our decisions get watered down by risk-averse committees practicing “Success Theater.” You need a blueprint to cut through the red tape, shift from reactive to proactive care, and build an agile, human-centric discovery engine.
Today, we are breaking down the essential strategies to rewire your R&D organization for speed, precision, and scale.
Transitioning from flawed animal models to 3D organoids and AI.
Using synthetic data to bypass crippling privacy bottlenecks.
Dismantling consensus culture to empower autonomous, fast-moving teams.
Let’s get right into the insights that are reshaping the industry.
If you’re a Pharma R&D Executive tired of watching your clinical timelines stretch while competitors accelerate, then here are the resources you need to dig into to rebuild your pipeline architecture:
Weekly Resource List:
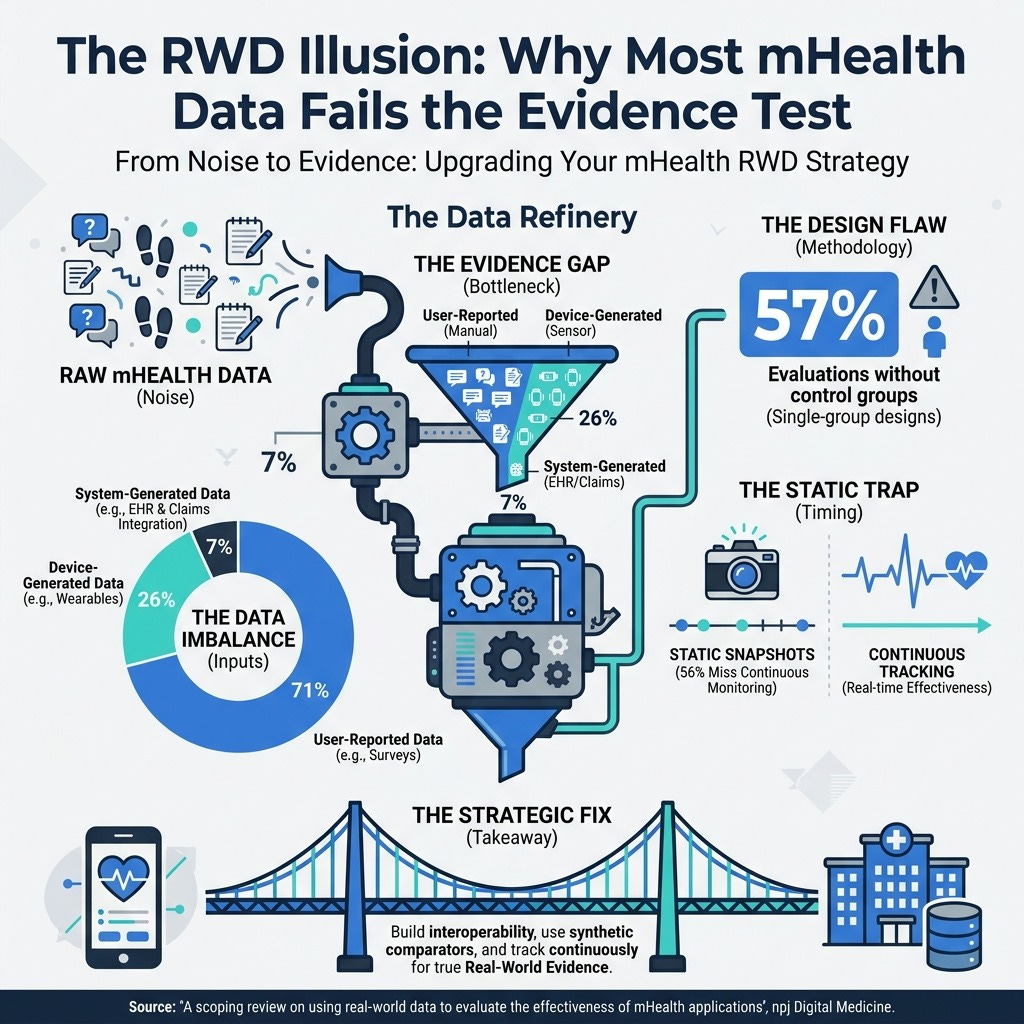
A scoping review on using real-world data to evaluate the effectiveness of mHealth applications | npj Digital Medicine (6 min read) - Most mHealth data fails the evidence test. 57% of evaluations lack control groups and 71% rely on subjective user inputs. The takeaway: You must build foundational interoperability and continuous tracking into your digital tools from day one to generate true Real-World Evidence.
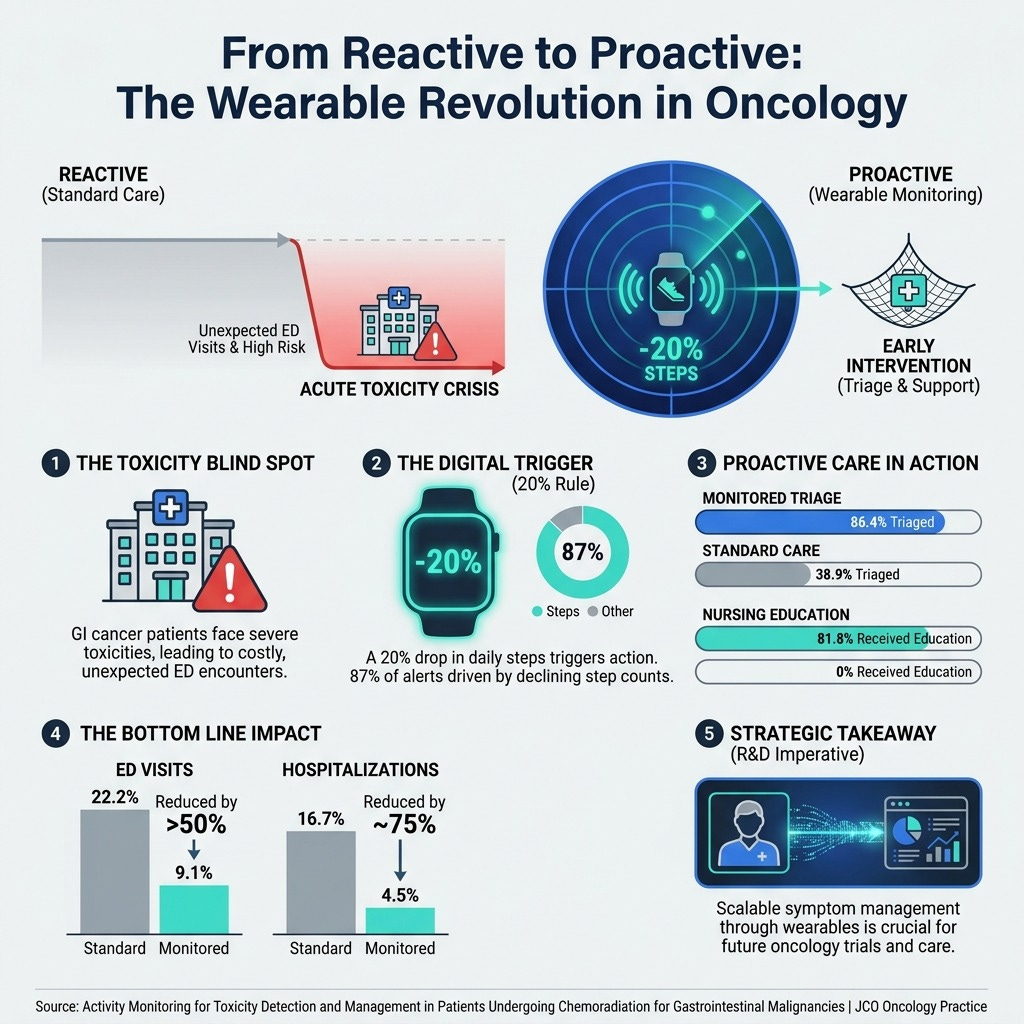
Activity Monitoring for Toxicity Detection and Management in Patients Undergoing Chemoradiation for Gastrointestinal Malignancies | JCO Oncology Practice (8 min read) - A simple 20% drop in daily steps from a smartwatch can predict severe treatment toxicity. Integrating passive activity data shifts cancer care from reactive emergency room visits to early, proactive outpatient triage.
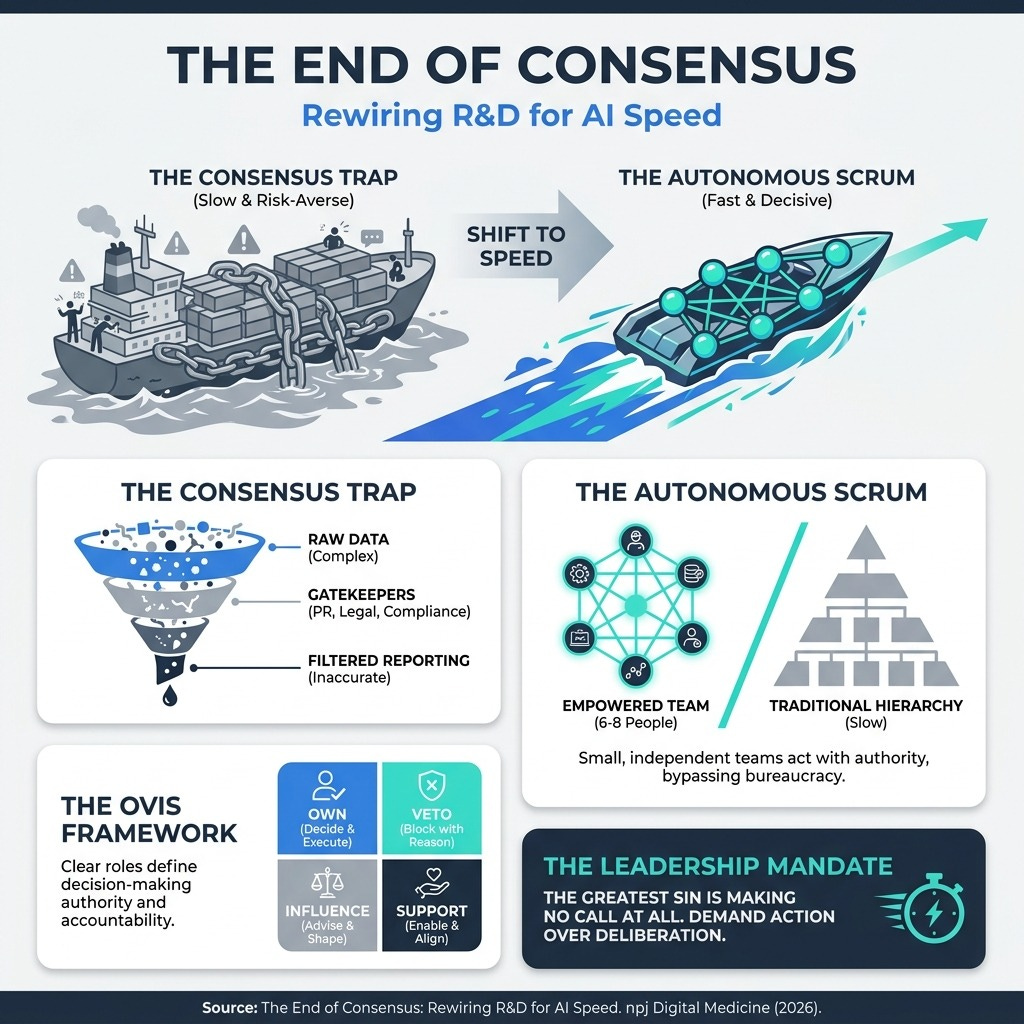
Decision-Making by Consensus Doesn’t Work in the AI Era | Harvard Business Review (7 min read) - Consensus management optimizes for risk mitigation, not speed, leaving the C-suite blind to reality. The takeaway: Implement the OVIS framework (Own, Veto, Influence, Support) and empower 6-to-8-person “Autonomous Scrums” to act immediately.
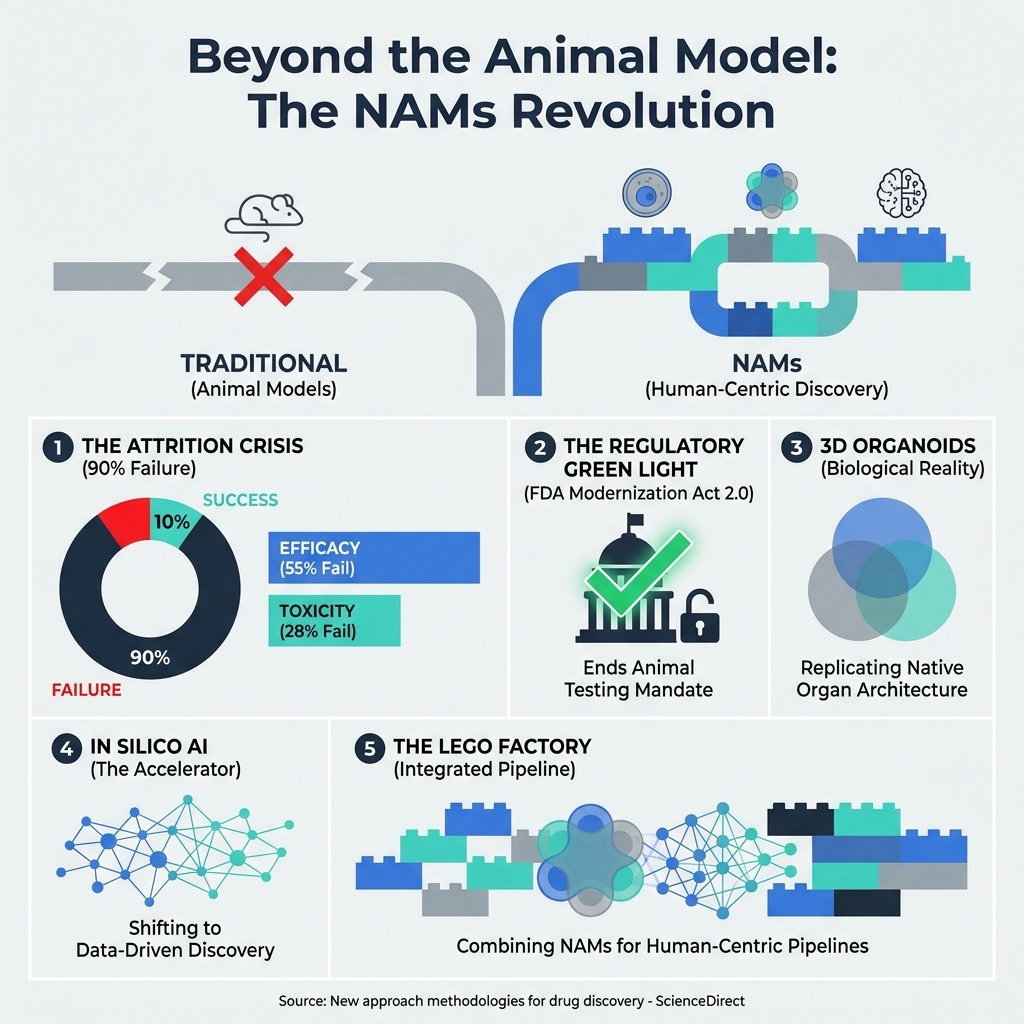
New approach methodologies for drug discovery | ScienceDirect (10 min read) - Over 90% of clinical drugs fail, largely due to our reliance on non-human animal models. The takeaway: The FDA Modernization Act 2.0 paves the way for human-centric New Approach Methodologies (NAMs). Combine AI, patient-specific stem cells, and 3D organoids to predict efficacy accurately.
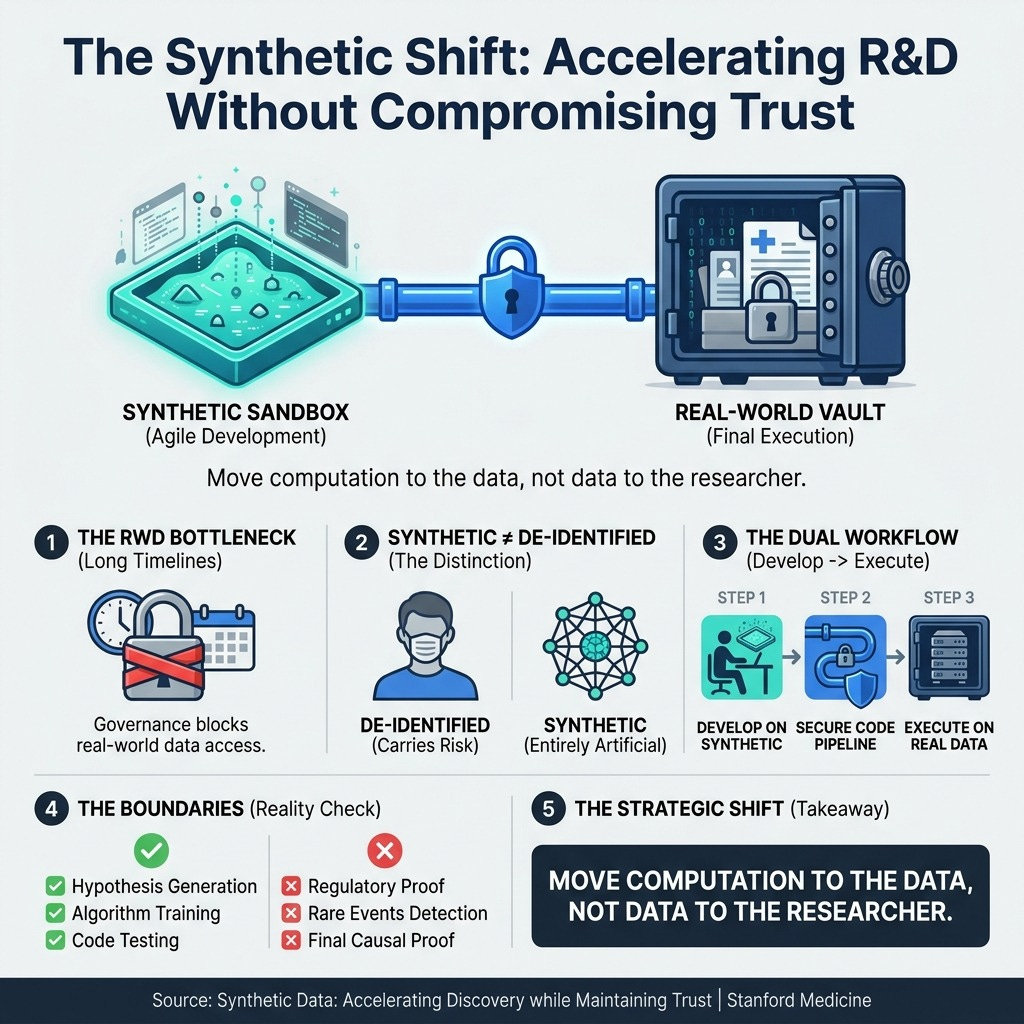
Synthetic Data: Accelerating Discovery while Maintaining Trust | Stanford Medicine (5 min read) - Privacy governance is stifling Real-World Data research. The takeaway: Stop moving data to researchers. Use entirely artificial synthetic datasets as a “sandbox” for rapid pipeline development before executing final code securely on real patient records.
3 Things To Accelerate R&D Innovation Even If Your Infrastructure is Slow
In order to achieve high-velocity, human-centric drug development, you’re going to need a handful of things.
Let’s break down exactly how you can implement these strategic shifts this week.
1. Swap Consensus for the OVIS Framework
You need to eliminate the ambiguity of committee decision-making and assign clear, absolute decision rights to small teams.
Consensus management creates “Success Theater,” where critical data is smoothed over by middle management to protect the status quo. By the time a report reaches your desk, the weak signals you need to make agile decisions are gone. To move at the speed of AI, you must adopt the OVIS model. Assign one person who Owns the decision, two or three who can Veto or Influence it, and require everyone else to Support it. Give your cross-functional scrums (groups of 6 to 8 people) the explicit authority to act, not just recommend. Speed is your new ultimate defensibility.
2. Build the “Lego Factory” of NAMs
You need to stop relying on flawed animal models and embrace a modular, human-centric discovery pipeline.
With clinical trial failure rates exceeding 90%, it is overwhelmingly clear that animal models cannot reliably predict complex human outcomes. You need to combine computational models (AI) with biological realities (3D organoids and stem cells). Treat these New Approach Methodologies like interchangeable Lego blocks. Use AI to forecast toxicity and generative drug design computationally, and then validate those findings using human vascularized organoids. This effectively future-proofs your pipeline against late-stage clinical attrition while aligning perfectly with the FDA Modernization Act 2.0.
3. Treat Synthetic Data as Your Development Sandbox
You must move computation to the data, rather than trying to move sensitive patient data to your researchers.
Waiting for compliance and privacy approvals on Real-World Data kills your team’s momentum. Instead, give your researchers access to high-fidelity synthetic data—which carries zero re-identification risk because it contains entirely artificial records that mimic real statistical structures. Instruct your data scientists to define their cohorts, test their machine learning models, and refine their analytical pipelines entirely in this synthetic sandbox. Once the pipeline is perfected, you can securely run the finalized code in the “vault” of real-world patient data. This accelerates discovery while maintaining absolute patient trust.
PS...If you’re enjoying Digital Health & AI News, please consider referring this edition to a friend. You’ll get exclusive access to our quarterly AI integration benchmarking report for making a referral.
And whenever you are ready, there are 2 ways I can help you:
The AI-Augmented Leader Email Course: Sign-up for my free 5-day email course on how to become an AI Augmented Leader in Lifesciences.
Strategic Roadmap Design: Translate your priorities across different parts of the organization into a coordinated and clear roadmap in 2026. Book time on my calendar to discuss this further.


Great read