Stop building engines without paved roads
AI won't fix your pipeline until you fix your data, culture, and tech debt.
HT4LL-20260507
Hey there,
We’ve spent billions on AI, yet it still takes 8.5 years to bring a drug to market because we are focusing on building a faster engine instead of laying down the right data roads.
In our rush to innovate, we often gravitate toward complex trial endpoints, hoping new technology will be the magic bullet. But behind the scenes, we’re drowning in data entropy, losing clinical sites to overly complicated protocols, and trapping our brightest leaders in an “identity lock” where they fail to scale their expertise. If we truly want to slash that stagnant clinical development timeline that hasn’t changed in 25 years, we have to stop treating AI as a shiny standalone object. We must do the unglamorous work of end-to-end workflow integration, prioritizing human sense-making, and fundamentally restructuring our disconnected data.
Today, we look at what’s actually working in modern drug development, sharing insights gathered from the Swiss Biotech Day held in Basel on May 4th & 5th.
Transitioning clinical data from rigid spreadsheets into semantic webs.
Escaping the “pilot purgatory” of AI in the lab by reducing technical debt.
Why your project managers are the real linchpins of trial execution.
Let’s dive right into how we can untangle this mess.
Swiss Biotech Day Panels:
Optimizing digital endpoint strategies for success: Are we overcomplicating clinical trials at the expense of our patients and data quality? To stop patients and sites from dropping out, we must simplify our trial endpoints. Consumer tech like an off-the-shelf smart watch can’t replace dedicated clinical patches when you need absolute data integrity. Ultimately, the true “lubricant” of trial execution is your Project Managers, who build the necessary trust and transparency when roadblocks hit.
Your pipeline isn’t the bottleneck - your organization is: Learning velocity as the hidden value driver in Swiss Biotech: Are biotechs prioritizing the speed of execution over the speed of sense-making? True learning velocity is the distance between insight and action. However, a constant “sense of urgency” is overrated; creativity actually happens when organizations give their people time to breathe, step away from their devices, and make sense of their work.
AI-powered pharma R&D, end-to-end: Connecting data, labs and decision: Why do so many promising AI pilots fail to scale? Most pilots fail not because of the technology, but because organizations ignore end-to-end integration and human workflows. For example, digital lab tools must accommodate real-world friction, like offering voice dictation for scientists whose hands are full with pipettes.
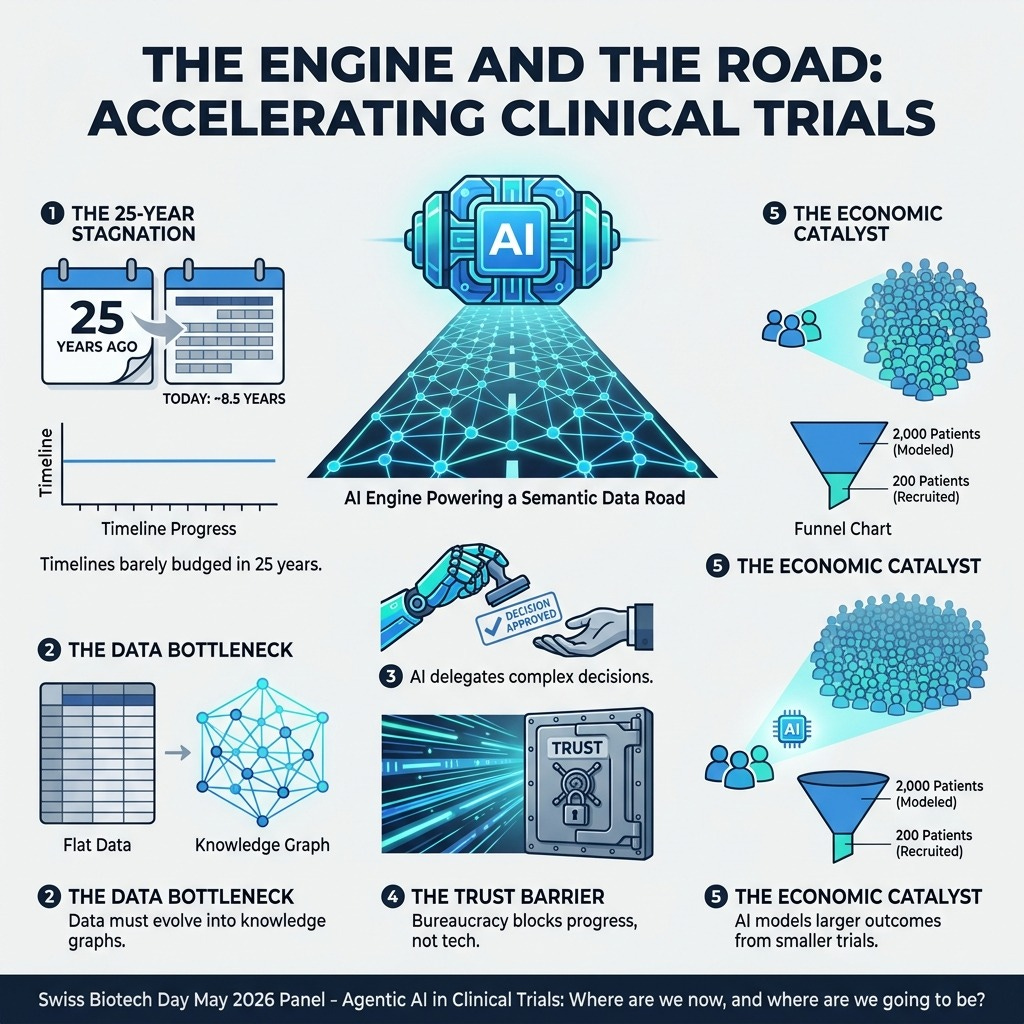
Agentic AI in clinical trials: Where are we now, and where we going to be? We often blame a lack of technological innovation for why it still takes nearly a decade to bring a drug to market, but the truth is, the powerful AI tools needed to accelerate the process are already here. The real barriers are outdated data infrastructures and a systemic lack of regulatory trust.
Transitioning from simple predictive tools to “agentic AI” that can automate complex decisions offers a massive opportunity to lower patient burden, reduce capital waste, and shorten trial durations. However, capitalizing on this requires biopharma leaders to shift their focus away from the AI “engine” itself and prioritize building the foundational data “road” it needs to operate.
4 Core Strategies to Accelerate Your Pipeline With AI
In order to truly transform your clinical operations and bridge the gap between scientific discovery and commercial reality, you’re going to need a handful of critical structural and cultural shifts.
1. Simplify Endpoints and Empower Your Human Linchpins
Are we overcomplicating clinical trials at the expense of our patients and data quality? You need to ruthlessly simplify your trial designs. Overly complicated protocols put a massive burden on clinical sites, which will simply choose to place patients on competing, simpler studies. When designing a patient-centric study, it is vital to balance scientific ambition with the actual human experience of the healthy volunteers or patients. Furthermore, while it is tempting to use consumer technology, an off-the-shelf smart watch or Amazon blood pressure cuff cannot replace dedicated clinical tools when you need absolute data integrity. To manage these complexities, your Project Managers must be empowered as the ultimate “lubricant in the machine”; they are the frontline defense for developing rapport with sites and ensuring rapid problem-solving when studies inevitably hit roadblocks.
2. Break the “Identity Lock” and Cultivate Learning Velocity
Are biotechs prioritizing the speed of execution over the speed of sense-making? You need to treat culture as a verb, not just a static framework. If your founders and top scientists are relying purely on their expert power, they will become a bottleneck—an “identity lock” that prevents the organization from successfully scaling its leadership. You must intentionally slow down; a constant “sense of urgency” is massively overrated. True creativity and learning velocity occur when you give teams the explicit time to step away, breathe, and make sense of their complex work. Finally, stop punishing failure. High-growth organizations adopt a “sometimes you win, always you learn” mentality, understanding that an open dialogue about failure is the fastest way to pivot toward commercial viability.
3. Escape Pilot Purgatory by Combating Data Entropy
Why do so many promising AI pilots in drug discovery fail to scale past the testing phase? You need to recognize that sitting on a massive amount of data means nothing if it lacks scientific context. Maintaining AI-ready data is a daily exercise, requiring consistent, meaningful annotations to ensure the organizational “data entropy” remains low over time. Additionally, you must guard against the hidden technical debt that accumulates during rapid, siloed AI proof-of-concepts. A pilot isn’t successful until it is deeply integrated into your scientists’ daily workflows, acting as a collaborative “co-scientist” that generates novel hypotheses to actively challenge—not just confirm—human intuition. Accelerate this adoption by openly publishing and analyzing your “good failure stories” instead of just the happy paths.
4. Pave Semantic Roads for Your Agentic AI Engines
Are we finally ready to break the 25-year stagnation in clinical trial timelines? You need to completely rethink how your clinical data is structured. Agentic AI has the power to automate complex decisions, moving beyond simple document generation, and potentially allowing us to run smaller trials by modeling larger populations. However, the most advanced AI engine is useless if it doesn’t have a properly structured data “road” to drive on. You must transition trial data out of traditional columns and rows and spin it up into connected semantic webs and knowledge graphs. This allows AI to accurately evaluate complex clinical dependencies. Widespread adoption won’t be gated by technology, but by overcoming organizational bureaucracy and building deep regulatory trust.
PS...If you're enjoying Healthtech for Lifescience Leaders, please consider referring this edition to a friend.
And whenever you are ready, there are 2 ways I can help you:
The AI-Augmented Leader Email Course: Sign-up for my free 5-day email course on how to become an AI Augmented Leader in Lifesciences.
Strategic Roadmap Design: Translate your priorities across different parts of the organization into a coordinated and clear roadmap in 2026. Book time on my calendar to discuss this further.